Introduction
At the heart of every chemical plant lies a set of reactors where raw materials are transformed into valuable products. From producing fuels and fertilizers to pharmaceuticals and polymers, chemical reaction engineering (CRE) plays a vital role in designing, analyzing, and optimizing these chemical reactors.
For chemical engineers, mastering the fundamentals of reaction engineering and reactor design is crucial. It allows them to scale laboratory reactions to industrial plants, ensure efficiency and safety, and minimize costs and environmental impact.
This article offers a comprehensive guide to the basics of chemical reaction engineering, focusing on reaction kinetics, reactor types, design equations, and real-world applications.
What is Chemical Reaction Engineering?
Chemical reaction engineering is the branch of chemical engineering that deals with the study of reaction kinetics, reactor modeling, and design. It provides the framework for:
- Predicting reaction rates and conversions.
- Designing reactors to achieve desired yields and selectivity.
- Scaling up from laboratory to industrial production.
- Optimizing energy consumption and safety.

Fundamentals of Reaction Kinetics
1. Rate of Reaction
The rate of reaction (r) describes how quickly reactants are consumed or products are formed.
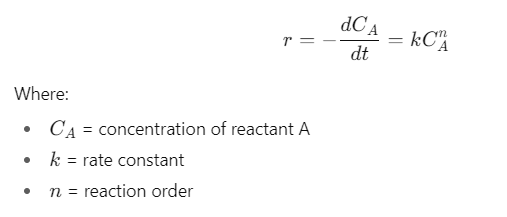
2. Reaction Order
- Zero order: Rate independent of concentration (r = k).
- First order: Rate proportional to one reactant (r = CA).
- Second order: Rate depends on square of concentration or two reactants (r = kCA2 or r = kCACB).
3. Arrhenius Equation
The rate constant depends on temperature:
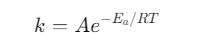
Where:
- A = frequency factor
- Ea = activation energy
- R = universal gas constant
- T = temperature
This explains why reactions accelerate at higher temperatures.
Types of Chemical Reactors
Reactors are classified based on flow characteristics and mixing behavior.
1. Batch Reactor
- Operates with fixed volume and no inflow/outflow during reaction.
- Widely used in labs and pharmaceuticals.
- Governing equation:
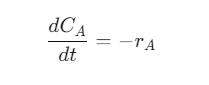
2. Continuous Stirred Tank Reactor (CSTR)
- Continuous inflow and outflow with complete mixing.
- Used in liquid-phase reactions.
- Design equation:
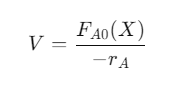
3. Plug Flow Reactor (PFR)
- Reactants flow through a tubular reactor with no back mixing.
- Suitable for large-scale continuous operations.
- Design equation:
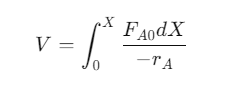
4. Packed Bed Reactor (PBR)
- Similar to PFR but filled with catalyst pellets.
- Used for gas-phase catalytic reactions.
5. Fluidized Bed Reactor
- Catalyst particles are suspended in fluid stream.
- Offers excellent heat transfer and mixing.
Reactor Design Equations
General Mole Balance
For species A in a reactor:
In – Out + Generation = Accumulation
Depending on reactor type, the accumulation term varies.
Multiple Reactions: Selectivity and Yield
In industry, reactions often involve parallel, series, or complex pathways. The design challenge is to maximize desired product yield while minimizing by-products.
- Selectivity (S): Ratio of desired product formed to undesired product.
- Yield (Y): Ratio of desired product formed to theoretical maximum.
Reactor choice and operating conditions (temperature, pressure, residence time) are key to controlling selectivity.
Heat Effects in Reactors
Reactions are either:
- Exothermic (release heat, e.g., combustion).
- Endothermic (absorb heat, e.g., cracking).
Temperature control is crucial for safety and efficiency. Reactors often require cooling jackets, heat exchangers, or quenching systems.
Non-Ideal Reactors
Real reactors deviate from ideal models due to:
- Dead zones (stagnant areas).
- Channeling (preferential flow paths).
- Axial mixing.
Models like Residence Time Distribution (RTD) and dispersion models are used to characterize non-ideal behavior.
Reactor Safety Considerations
- Thermal Runaway: Exothermic reactions may accelerate uncontrollably.
- Pressure Buildup: Gas evolution may exceed venting capacity.
- Catalyst Deactivation: Fouling, poisoning, or sintering reduces activity.
Process safety requires inherent safety design, relief systems, and emergency protocols.
Applications of Chemical Reactors
Ammonia Synthesis (Haber-Bosch Process)
- High pressure PBR with iron catalyst.
Polymerization Reactors
- CSTRs for polyethylene and polypropylene.
Petroleum Refining
- Fluidized catalytic cracking (FCC).
Pharmaceuticals
- Batch reactors for precision and flexibility.
Biochemical Processes
- Fermenters for antibiotics, enzymes, and ethanol.
Reactor Design Workflow
- Define process requirements (conversion, selectivity).
- Gather kinetic data (lab experiments).
- Choose reactor type (batch, CSTR, PFR).
- Apply design equations.
- Consider heat/mass transfer effects.
- Validate with pilot plant.
- Scale up to industrial size.
Role of Computational Tools
Modern design uses simulation software:
- Aspen Plus / Aspen HYSYS – Reactor modeling.
- COMSOL Multiphysics – CFD analysis of mixing/heat transfer.
- MATLAB/Python – Custom kinetic modeling.
Case Study: Ammonia Plant Reactor Design
- Reaction: N₂ + 3H₂ ⇌ 2NH₃ (exothermic).
- Reactor Type: Multi-bed PBR with intercoolers.
- Design Considerations: High pressure (150–250 bar), temperature (400–500°C), equilibrium limitations, and catalyst life.
Outcome: Optimized design yields 15–20% per pass conversion, with recycle for efficiency.
Challenges in Reactor Design
- Scaling up from lab to industrial scale.
- Non-ideal flow patterns.
- Handling multiphase systems.
- Catalyst deactivation and regeneration.
- Balancing economics with safety.
Future of Reactor Design
- Process Intensification: Microreactors for safer, faster reactions.
- Green Chemistry: Designing reactors for minimal waste and energy use.
- Digital Twins: Real-time reactor monitoring and predictive modeling.
- AI & Machine Learning: Optimizing operating conditions dynamically.
Conclusion
Chemical reaction engineering lies at the core of chemical engineering practice. By understanding kinetics, reactor types, and design principles, engineers can scale up reactions, optimize yields, and ensure safety. Reactor design is both a science and an art, requiring integration of thermodynamics, transport phenomena, and process safety.
As industries push toward sustainability and digitalization, the role of chemical reaction engineers becomes even more critical. Mastering these fundamentals prepares engineers to innovate and lead in building safer, greener, and more efficient chemical plants.