Introduction
At the very heart of chemical engineering lies the discipline of transport phenomena, a subject that unifies the principles of momentum transfer (fluid mechanics), heat transfer, and mass transfer. These three pillars describe how momentum, energy, and matter move through physical systems. Whether it’s designing a heat exchanger, modeling pollutant diffusion, or predicting flow through a packed column, transport phenomena provide the fundamental tools for analysis and design.
This comprehensive article explores the fundamentals of transport phenomena, their governing laws, key equations, real-world applications, and relevance in modern chemical engineering.
What Are Transport Phenomena?
Transport phenomena describe the mechanisms by which momentum, heat, and mass move within and between systems. It is a broad and unifying field that connects physics, chemistry, and engineering.
- Momentum transfer (fluid mechanics): Flow of fluids and the forces driving motion.
- Heat transfer: Energy movement due to temperature differences.
- Mass transfer: Movement of chemical species driven by concentration differences.
These are not isolated domains; in chemical processes, they often occur simultaneously.



Applications
- Pump and compressor design
- Pressure drop in pipelines
- Fluidized bed reactors
- Aerodynamics in combustion systems
2. Heat Transfer
Heat transfer is the movement of energy due to a temperature difference.

Dimensionless Numbers in Heat Transfer
- Nusselt Number (Nu): Ratio of convective to conductive heat transfer.
- Prandtl Number (Pr): Ratio of momentum diffusivity to thermal diffusivity.
Applications
- Heat exchanger design (shell-and-tube, plate type)
- Reactor cooling jackets
- Boiler and furnace design
- Cryogenic processes
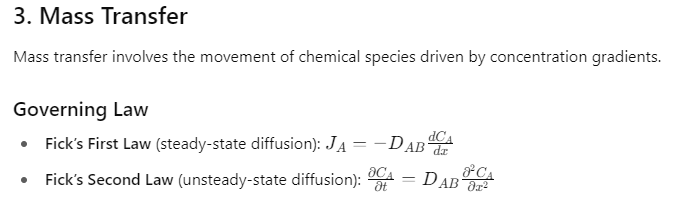
Dimensionless Numbers in Mass Transfer
- Reynolds Number (Re): Flow regime indicator
- Schmidt Number (Sc): Ratio of momentum diffusivity to mass diffusivity
- Sherwood Number (Sh): Dimensionless mass transfer coefficient
Applications
- Distillation, absorption, extraction
- Drying of solids
- Gas absorption in scrubbers
- Membrane separations (RO, NF, UF)
Interplay of Momentum, Heat, and Mass Transfer
In real-world systems, transport phenomena rarely occur in isolation:
- Evaporation: Heat transfer drives mass transfer.
- Cooling towers: Heat and mass transfer occur simultaneously.
- Combustion: Momentum, heat, and mass interact strongly.
- Multiphase reactors: Complex coupling of all three phenomena.
Dimensionless Analysis and Similarity
Transport phenomena rely heavily on dimensionless groups to generalize results across systems:
- Reynolds Number (Re) – Fluid flow behavior
- Prandtl Number (Pr) – Thermal boundary layer characteristics
- Schmidt Number (Sc) – Mass transfer analog of Prandtl
- Nusselt Number (Nu) – Heat transfer efficiency
- Sherwood Number (Sh) – Mass transfer efficiency
- Grashof Number (Gr) – Natural convection driving force
These groups enable scaling and similarity analysis, vital for moving from lab-scale experiments to industrial applications.
Tools and Simulation in Transport Phenomena
Modern engineers use computational tools to model transport problems:
- CFD (Computational Fluid Dynamics): ANSYS Fluent, OpenFOAM
- Process Simulators: Aspen Plus, COMSOL Multiphysics
- Programming: MATLAB, Python for custom models
These allow visualization of velocity profiles, temperature gradients, and concentration fields.
Real-World Applications in Industries
Chemical and Petrochemical Industries
In chemical and petrochemical plants, transport phenomena are central to the design and operation of reactors, distillation columns, absorbers, and extraction units. Mass transfer principles are harnessed for separation techniques such as distillation, absorption, crystallization, and liquid–liquid extraction, enabling the efficient purification of products and removal of impurities. Additionally, heat conduction (via heat exchangers and reactors) and momentum transfer (fluid flow in pipelines and mixing tanks) are optimized to maximize yield and maintain safety and process stability.
Food and Pharmaceutical Industries
Transport phenomena guide critical processes in food and pharmaceutical manufacturing. In the food sector, heat and mass transfer analysis is vital for pasteurization, drying, freezing, and packaging, which all require precise temperature and moisture control to ensure quality and safety. Pharmaceutical production relies on mass transfer for drug formulation and targeted drug delivery, where diffusion and convection mechanisms control the release and transport of active compounds within biological systems.
Water and Environmental Engineering
Transport phenomena are extensively applied in water treatment and environmental pollution control. In water purification plants, mass transfer enables contaminant removal through filtration, membrane processes, or adsorption. Environmental engineers use fluid dynamics and dispersion models to predict the movement of pollutants in air, soil, or water, supporting remediation strategies and regulatory compliance.
Advanced Manufacturing and Energy Systems
Modern manufacturing techniques, such as microfluidics and additive manufacturing (3D printing), depend on precise control of mass and heat transfer at small scales for rapid prototyping and material synthesis. Energy systems, including fossil fuel and renewable energy (e.g., solar thermal plants), utilize transport phenomena to optimize heat recovery, manage waste heat, and improve system efficiency.
Metallurgy and Materials Processing
In metallurgy, transport phenomena govern the solidification, casting, and refinement of metals and alloys. Control over heat transfer during smelting and solidification ensures desirable microstructures and properties in the final product. The principles are also pivotal in processes such as chemical vapor deposition and crystal growth for electronics and advanced material fabrication.
Environmental Safety and Sustainability
Transport phenomena help monitor and minimize industrial emissions, design safer processes, and develop cleaner technologies. Examples include the modeling of pollutant dispersion to mitigate environmental impacts and the design of equipment for efficient resource and energy use, supporting circular economy initiatives and regulatory compliance.
In summary, transport phenomena provide a foundational toolkit for understanding, modeling, and improving virtually every industrial process involving the movement of mass, energy, or momentum, supporting innovation and sustainability across global industries.
Educational Importance
Transport phenomena is a cornerstone subject in chemical engineering education. It trains students to:
- Build physical intuition of how systems behave
- Solve differential equations for practical problems
- Connect micro-level molecular behavior to macro-scale plant operations
Common textbooks include:
- “Transport Phenomena” by Bird, Stewart, and Lightfoot
- “Fundamentals of Momentum, Heat, and Mass Transfer” by Welty et al.
Challenges in Mastering Transport Phenomena
- Heavy reliance on mathematics and differential equations
- Abstract nature of the subject
- Coupling between momentum, heat, and mass transfer makes real systems complex
- Bridging theory with industrial practice
The Future of Transport Phenomena
- Microfluidics: Transport in lab-on-chip devices
- Nanotechnology: Mass and heat transport in nanostructures
- Renewable Energy: Heat and mass transfer in solar cells, fuel cells
- AI Integration: Machine learning models for transport predictions
Conclusion
Transport phenomena form the bedrock of chemical engineering. By mastering the principles of momentum, heat, and mass transfer, engineers can design safer, more efficient, and more sustainable processes. From the smallest microchannel to the largest industrial reactor, transport phenomena guide how materials and energy move.
In a world increasingly focused on efficiency and sustainability, understanding transport phenomena is not just academic—it’s a professional necessity.
Final Thought: Master transport phenomena, and you master the language of chemical engineering itself.